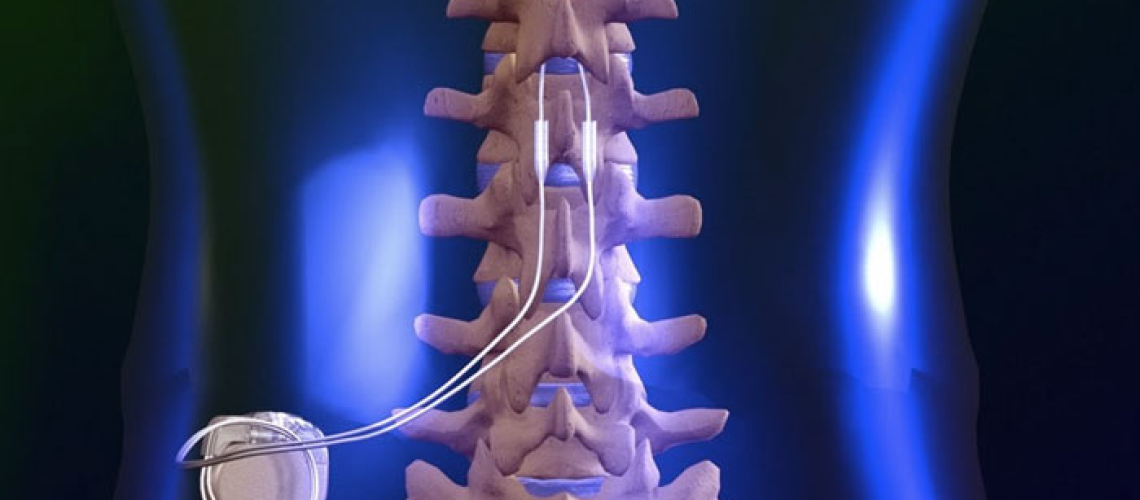
How a Spinal Cord Stimulator Lawsuit Could Help
For individuals who have been diagnosed with an injury linked to use of a spinal cord stimulator, the potential for legal recourse is a critical consideration. Victims may be entitled to financial compensation to cover the following:
- Medical expenses
- Lost wages
- Pain and suffering
- Punitive damages